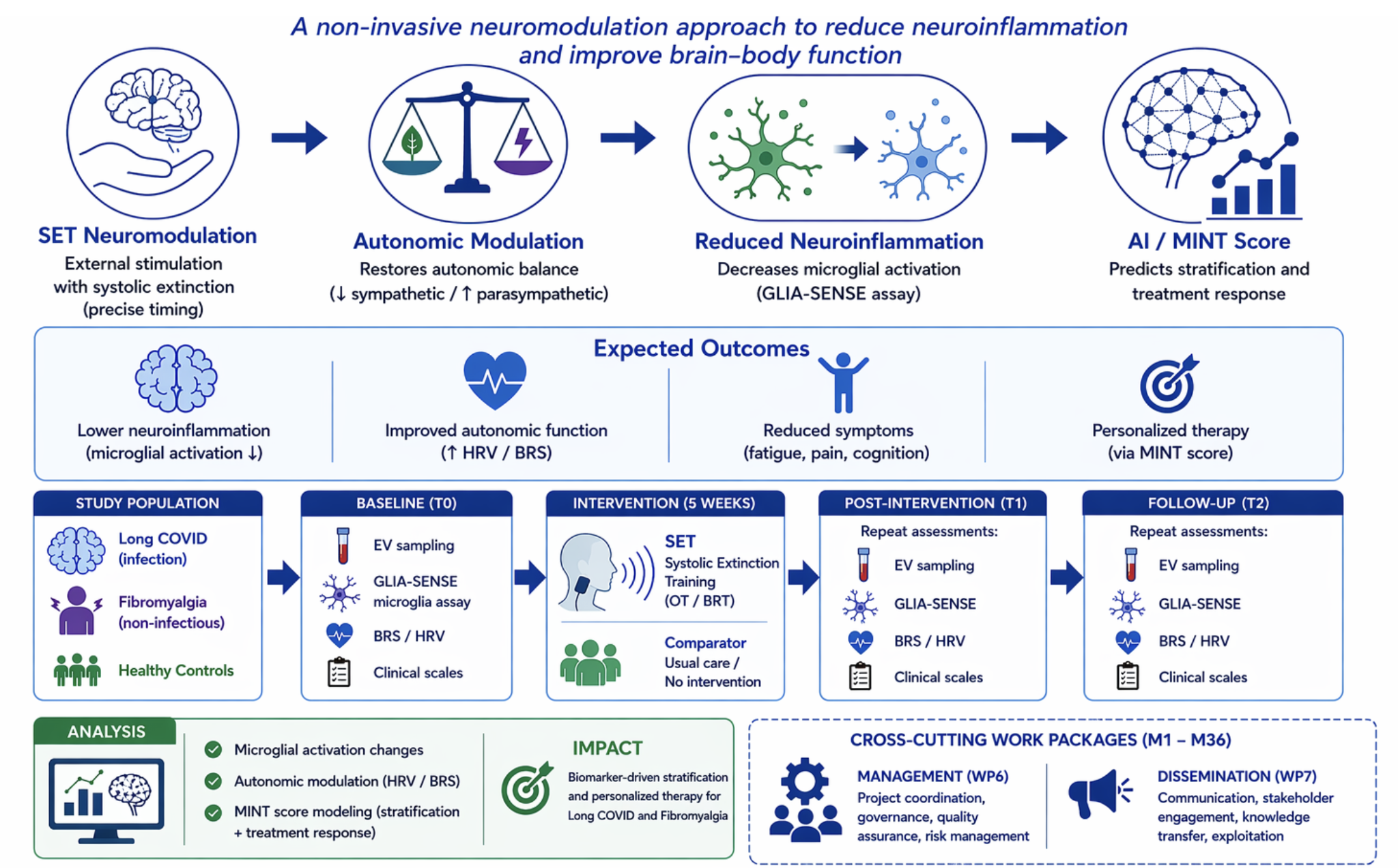
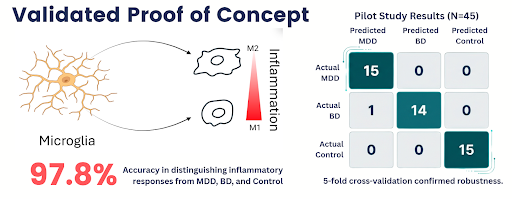
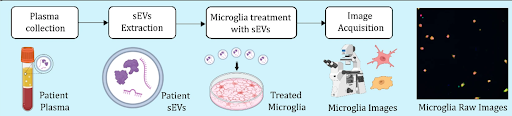
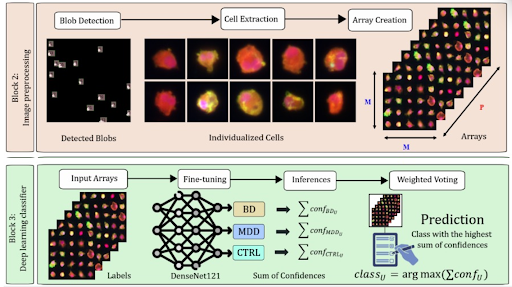
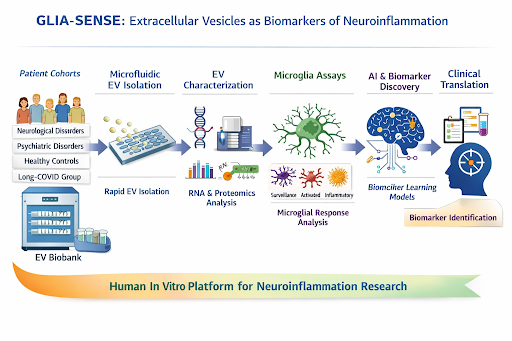
InBiomics is a pioneering medical research and technology project that will redefine the diagnostic and therapeutic landscape for post-infectious chronic conditions like Long COVID, Pain, and Psychiatric disease. The project centers on the GLIA-SENSE Platform, which utilizes human microglia as biological sensors to "read" a patient’s neuroinflammatory state from a routine blood draw. The Mission is to replace subjective, symptom-based classifications with objective, mechanism-validated biomarkers—specifically the MINT Score (Microglial Inflammatory Neuro-Trajectory)—to guide precise clinical decision-making. The platform integrates advanced microfluidics for isolating patient-derived extracellular vesicles (EVs) with AI-driven high-content imaging to decode complex inflammatory signals. Furthermore, the project validates Systolic Extinction Training (SET), a non-invasive neuromodulation therapy designed to restore autonomic balance. By industrializing transportable lab-on-chip modules and standardized "Starter Kits," Long_COVID_Impuls will make high-precision functional profiling feasible and affordable (~€29 per sample), enabling a decentralized network of specialized clinics to end the "diagnostic odyssey" for millions of patients suffering from post-infectious brain disorders.
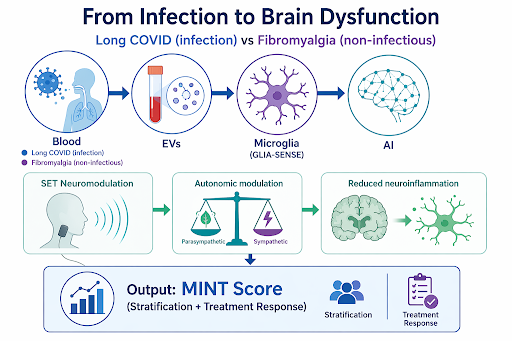
Figure 1: Platform diagram for microglial profiling and the SET device for targeted neuromodulation.
ITHE GLOBAL BURDEN OF POST-INFECTIOUS SEQUELAE. Post-infectious chronic conditions, exemplified by Long COVID, Chronic Pain, and Psychiatric disease, represent one of the most pressing unresolved challenges in contemporary medicine. The World Health Organization estimates that 10–20% of individuals who recover from acute SARS-CoV-2 infection develop persistent symptoms lasting months to years, including chronic pain, fatigue, cognitive impairment (“brain fog”), depression, and autonomic dysregulation. Across the European Union alone, conservative estimates suggest ≅30 million people have experienced or are living with post-COVID sequelae. Chronic pain — one of the cardinal symptoms — costs European healthcare systems over €200 billion annually in direct treatment and lost productivity. Yet for the individual patient, the reality is more immediate: months or years of unexplained suffering, repeated inconclusive investigations, and treatments that address symptoms without engaging the underlying disease process.
CRITICAL LIMITATIONS IN CURRENT DIAGNOSTIC PARADIGMS. Despite the scale of this challenge, diagnostic approaches remain fundamentally inadequate. Current clinical practice relies on symptom-based classification (e.g., WHO case definitions), which fails to capture the biological mechanisms driving persistent symptoms. Three classes of diagnostic tools are currently available, and each has critical limitations. Molecular biomarkers such as circulating cytokines (IL-6, TNF-α, CRP) lack specificity: they are elevated in hundreds of conditions and fail to distinguish post-infectious neuroinflammation from metabolic, autoimmune, or idiopathic states. Advanced neuroimaging such as TSPO-PET can detect microglial activation in vivo, but is expensive (€3,000–5,000 per scan), low-throughput, exposes patients to ionising radiation, and is available only at specialised research centres — entirely unsuitable for routine clinical use. Clinical diagnostics remain symptom-based, preventing mechanistic stratification and targeted treatment. As a result, patients endure what they describe as a “diagnostic odyssey”: cycling through specialists, receiving conflicting diagnoses, and receiving treatments chosen through trial and error rather than mechanism.
NEUROINFLAMMATION AND THE ROLE OF THE VAGUS NERVE. A growing body of evidence identifies neuroinflammation as a central mechanistic driver of post-infectious brain dysfunction. Histopathological studies have demonstrated viral RNA and inflammatory infiltrates in neural structures including the vagus nerve, indicating direct infection-mediated disruption of neuroimmune regulation (Woo et al., Acta Neuropathol 2023). The vagus nerve plays a dual role in this pathology. Through the inflammatory reflex (Andersson & Tracey, J Intern Med 2024), it constitutes the principal neural pathway by which the brain regulates systemic cytokine release: afferent signals carry peripheral inflammatory information to the nucleus tractus solitarius (NTS), and efferent cholinergic signals suppress macrophage activation. Disruption of this axis — through direct infection, inflammation, or chronic stress — initiates self-amplifying cycles of neuroinflammation and autonomic dysfunction. The structural properties of the vagus nerve — its partial myelination, high metabolic activity, and extensive inflammatory surface — make it a particularly vulnerable target in SARS-CoV-2 infection.
DIFFERENTIATING PATHOPHYSIOLOGY: LONG COVID VS. FIBROMYALGIA. Fibromyalgia provides a scientifically powerful comparator condition. It shares the symptom profile of Long COVID — chronic pain, fatigue, sleep disturbance, cognitive dysfunction — yet arises from non-infectious central sensitisation mechanisms characterised by sympathetic overactivation rather than post-infectious vagal impairment . Preliminary data from the ongoing BMBF-funded DATIPilot study (03DPS1256A) demonstrate that Long COVID and fibromyalgia exhibit distinct autonomic signatures: Long COVID is associated with parasympathetic (vagal) dysfunction and an increased very-high-frequency (vHF) component of heart rate variability (HRV), suggesting a pathological parasympathetic response to stress, along with heart rate values significantly above diastolic blood pressure — an expression of autonomic dysfunction. Fibromyalgia, in contrast, shows an increased sympathetic stress response (Thieme et al., J Pain 2015). Both conditions display diminished baroreflex sensitivity (BRS), but the underlying pathophysiology differs: in Long COVID, BRS reduction reflects vagal afferent damage; in fibromyalgia, it reflects learned sympathetic dominance from chronic stress.

